ASH 2025 B-Cell Lymphoma Updates
MCL, MZL, and Waldenström Macroglobulinemia
和信治癌中心腫瘤內科部
2025-12-01
Mantle Cell Lymphoma (MCL)
Agenda
Mantle Cell Lymphoma (MCL)
Marginal Zone Lymphoma (MZL)
Waldenström Macroglobulinemia (WM)
Clinical Implications
Novel CAR-T, BTKi Combinations, and Targeted Therapies
GLPG5101: Next-gen CAR-T with 7-day manufacturing
ATALANTA-1 trial (NCT06561425)
Background
- Fresh CAR-T with decentralized manufacturing 1
- Eliminates 3-6 week traditional manufacturing delay
Method
- N=24 MCL patients, single fixed IV dose
- 7-day median vein-to-vein time (96% achieved)
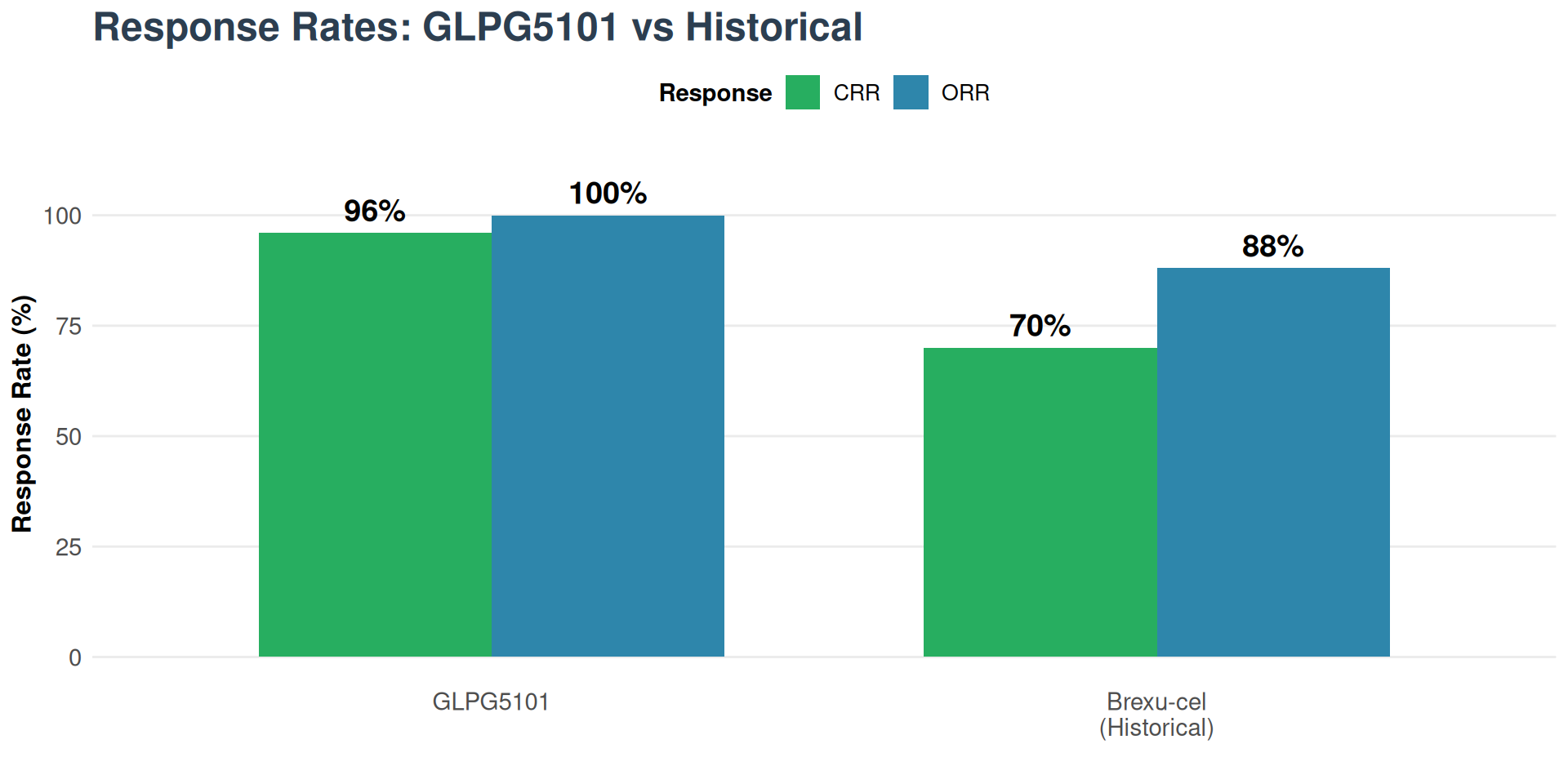
Results
- ORR: 100% (versus 85-91% brexu-cel historical)
- CRR: 96% (versus 59-82% brexu-cel)
- MRD negative: 90% in evaluable patients
- CAR-T persistence: Up to 21 months
GLPG5101: Safety Profile & Regulatory Status
Safety Profile
| Adverse Event | GLPG5101 | Brexu-cel |
|---|---|---|
| Grade ≥3 CRS | 0-4% | 8-15% |
| Severe Neurotoxicity | 4% | 31-32% |
| Bridging Chemo | Not required | Required |
- Most common AEs: hematologic (manageable)
- DOR and PFS: 83% at 9 months
- No treatment-related deaths
Regulatory Status
- FDA RMAT Designation: August 2025
- Technology proof-of-concept established
⚠️ Commercial Uncertainty
- Galapagos announced cell therapy wind-down: October 2025
- 365 employees affected across 5 facilities
- No viable acquisition offers received
- Future development uncertain despite promising data
Expert Opinions on GLPG5101
Clinical Investigators
- Marie Jose Kersten, MD, PhD from Amsterdam UMC said:
- “We saw deep responses including patients with high-risk features… a 96% CR with duration of response at 9 months of 83%.”
- “We would feel very comfortable to deliver this therapy on an outpatient basis.”
- Omotayo Fasan, MBBS from Galapagos said:
- “By initiating lymphodepletion immediately after cell collection, we observed a low 5% attrition rate, compared to rates of up to 30% reported in some clinical trials and real-world settings.”
Manufacturing Innovation
- Marie Jose Kersten on feasibility:
- “Our data show that it’s feasible to decentralize CAR T-cell manufacturing, and with a fresh-out/fresh-in procedure, you can have a very short vein-to-vein time.”
- Omotayo Fasan on T-cell phenotype:
- “Fresh, early memory CAR-T cells can be reliably delivered — this stem cell-like phenotype correlates with CAR-T persistence up to 21 months.”
⚠️ Commercial Update
- Henry Gosebruch (Galapagos CEO) on wind-down:
- “No viable proposals were received with terms or financing that would reasonably support the business’ future.”
First‐line treatment of mantle cell lymphoma.
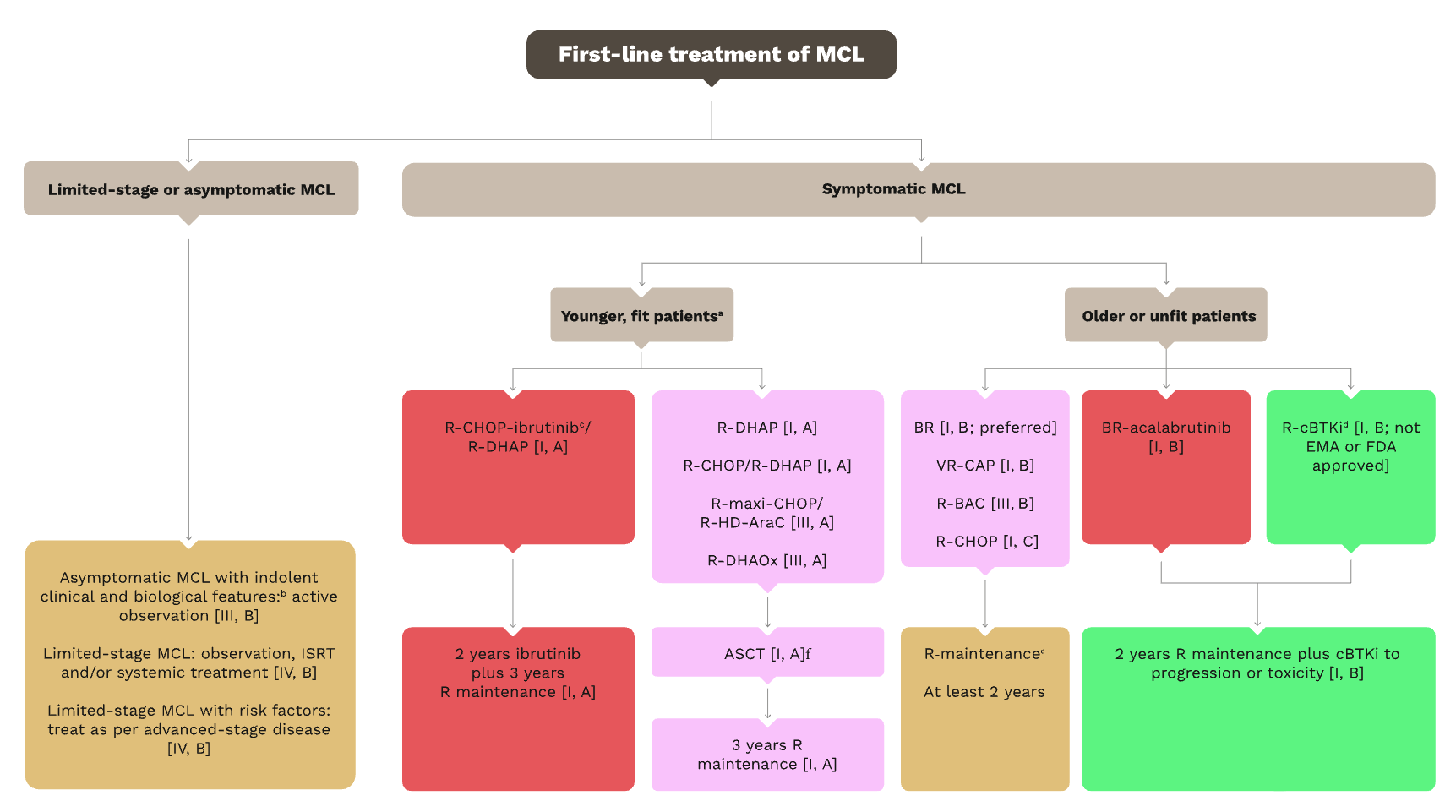
Caption: First‐line treatment of mantle cell lymphoma.
ECHO trial: Acalabrutinib + BR for frontline MCL
Phase III (NCT02972840)
Background
- Transplant-ineligible untreated MCL 1
- N=598 patients, median follow-up 50 months
Method
- Acalabrutinib 100mg BID + Bendamustine-Rituximab
- versus Placebo + BR
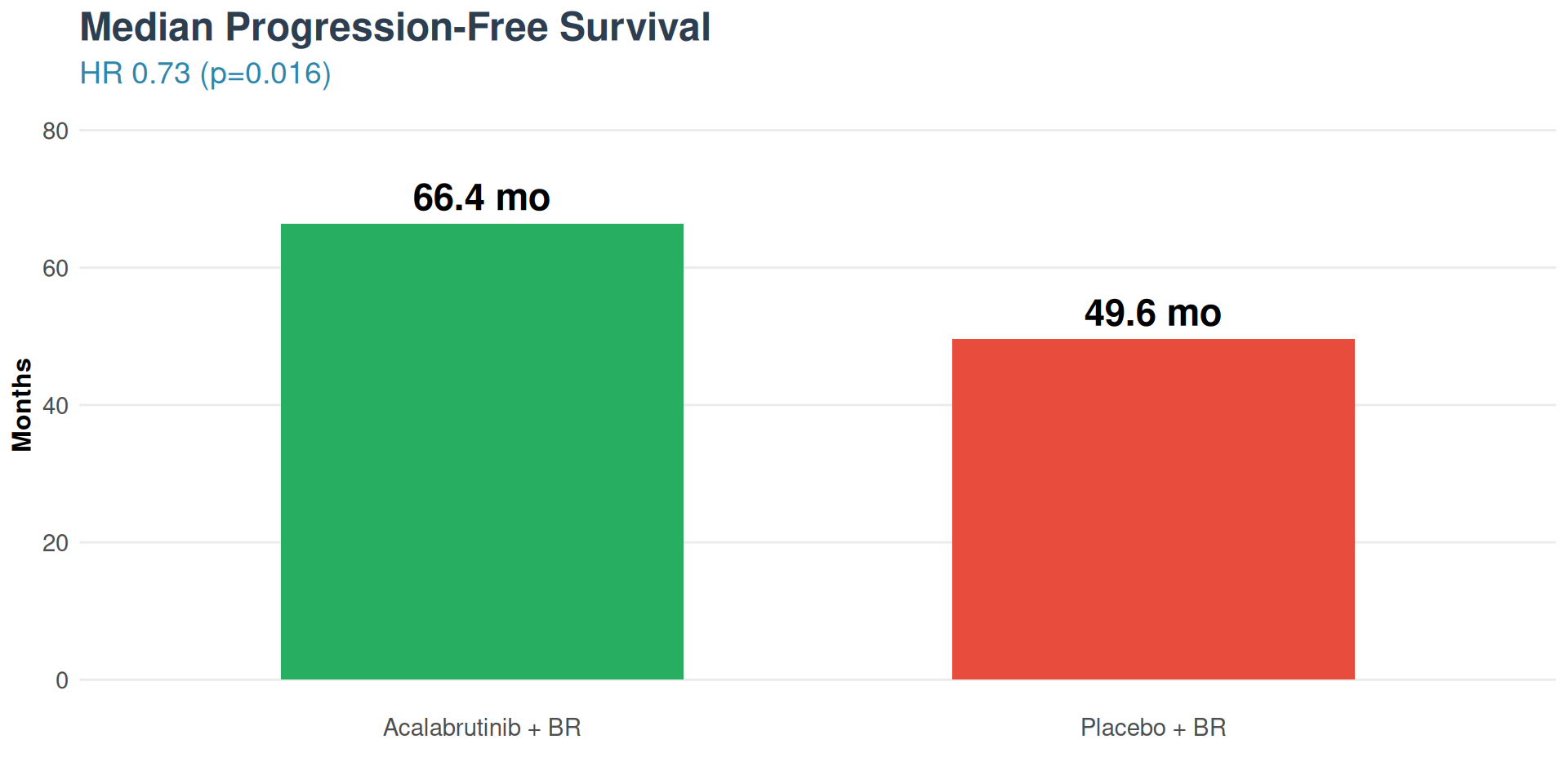
Results
- mPFS: 66.4 versus 49.6 months
- HR 0.73 (95% CI 0.57-0.94, p=0.016)
- 27% reduction in progression/death risk
- Ki-67 ≥30%: HR 0.69 (high proliferation benefit)
ECHO Trial: Safety & Regulatory
Additional Efficacy Data
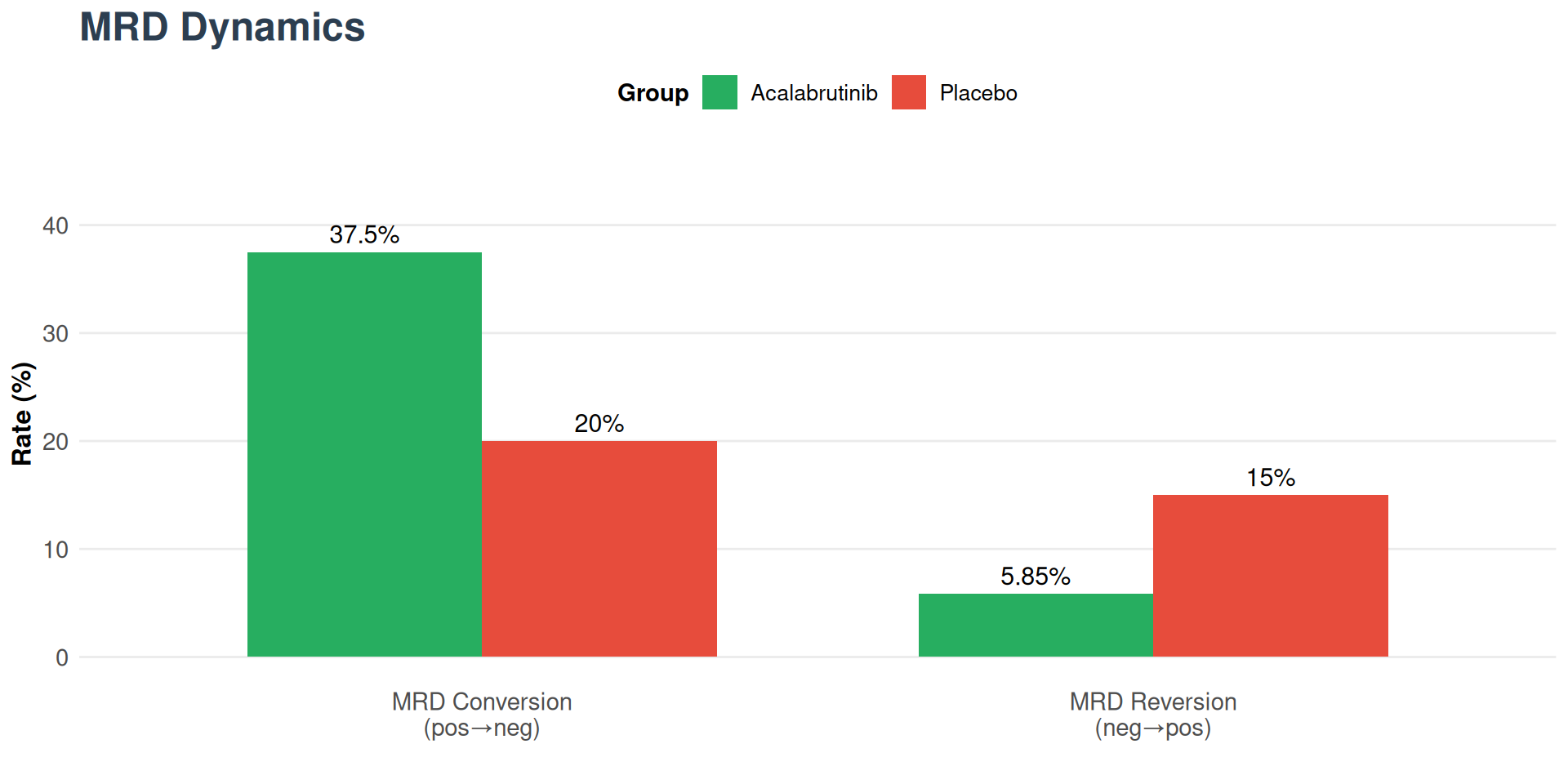
Regulatory Status
- FDA Traditional Approval: January 16, 2025
- Approved for transplant-ineligible untreated MCL
- Converted from accelerated approval
- EMA Approval: May 6, 2025
- Project Orbis international review collaboration
Subgroup Analysis
- TP53 mutant: HR 0.88 (modest benefit)
- ORR: 91% vs 88%
- CR rate: 66.6% vs 53.5%
PFS and OS with and without COVID-19 deaths and PFS by acalabrutinib exposure
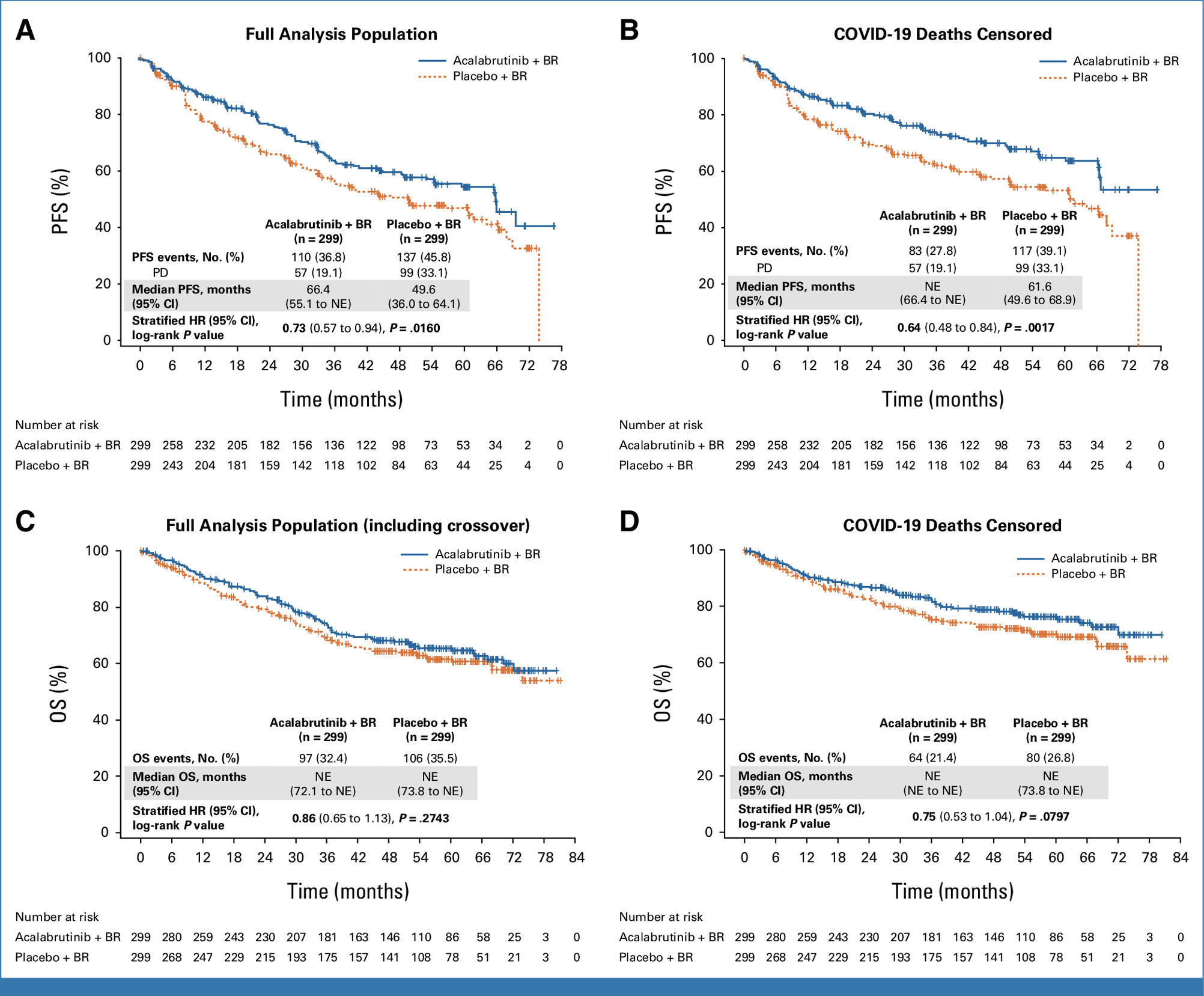
Wang M, Salek D, Belada D, et al. Acalabrutinib Plus Bendamustine-Rituximab in Untreated Mantle Cell Lymphoma. Journal of Clinical Oncology. 2025;43(20):2276-2284. doi:https://doi.org/10.1200/jco-25-00690
Expert Opinions on ECHO Trial
Lead Investigators
- Michael Wang, MD from MD Anderson Cancer Center said:
- “BR is no longer the standard of care. Acalabrutinib plus BR is the current standard for older patients with newly diagnosed MCL.”
- “I believe that we’re in the process of curing many people.”
- Martin Dreyling, MD from LMU Munich said:
- “For the vast majority of MCL patients, now we have the option to treat with an on-label BTK inhibitor plus chemotherapy.”
- “BTK inhibitors are a mandatory part of first-line treatment.”
Clinical Perspectives
- Tycel Phillips, MD from City of Hope said:
- “[The trial] did meet its primary endpoint, which was a PFS benefit, and there was a hint that there was some trend toward an OS benefit.”
- “Reserve the ECHO regimen for patients who had a more difficult disease to treat.”
- Martin Dreyling on tolerability:
- “[Acalabrutinib is] better tolerated, especially regarding the typical adverse effects like atrial fibrillation or bleeding disorders, both of which are especially relevant for older patients.”
Industry Response
- Susan Galbraith, MD from AstraZeneca said:
- “These impactful results in mantle cell lymphoma show that bringing acalabrutinib to the first-line setting significantly delays disease progression.”
BOVen: Chemo-free triplet for TP53-mutant MCL
Phase II (NCT03824483)
Background
- TP53-mutant MCL: historical mPFS 0.9y, mOS 1.8y 1
- N=25 TP53-mutant patients
Method
- Zanubrutinib + Obinutuzumab + Venetoclax
- MRD-guided discontinuation after 24 cycles
Efficacy Results
| Outcome | BOVen | Historical (Chemo) |
|---|---|---|
| ORR | 96% | 50-60% |
| CR | 88% | 30-40% |
| 2-year PFS | 72% | <20% |
| 2-year OS | 76% | ~40% |
| uMRD (10⁻⁵) | 95% | Rarely achieved |
BOVen: Safety & Guideline Update
Safety Profile
| Adverse Event | Rate |
|---|---|
| Diarrhea | 64% (mostly low-grade) |
| COVID-19 infection | 56% |
| Infusion reaction | 24% |
| Neutropenia | 32% |
| Grade 4 TRAEs | 0% |
- Time-limited therapy possible with MRD guidance
- No treatment discontinuation due to toxicity
Regulatory & Guidelines
NCCN Category 2A
BOVen now included for TP53-mutant MCL
- Published in Blood January 2025
- Expansion cohort: additional 25 patients enrolling
- Paradigm shift for high-risk MCL
- First effective chemo-free option for TP53-MCL
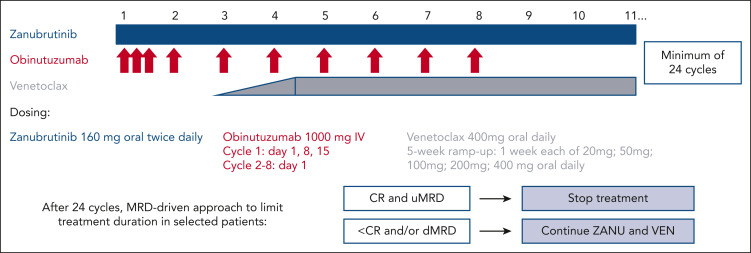
Expert Opinions on BOVen Trial
Lead Investigator
- Anita Kumar, MD from Memorial Sloan Kettering Cancer Center said:
- “TP53-mutant MCL is a high-risk entity with poor outcomes on standard chemo-immunotherapy. The idea was to develop biologically targeted therapies.”
- “This is the first dedicated study with reported outcomes specifically for this cohort, demonstrating that clinical trials are feasible.”
- “This two-year PFS of 72% was a huge improvement over historical data.”
Additional Expert Perspectives
- William B. Pearse, MD from UC San Diego said:
- “BOVen presents a crucial opportunity to explore how BTK inhibition combined with other targeted agents may serve as a chemotherapy-sparing approach.”
- Austin I. Kim, MD from Dana-Farber Cancer Institute said:
- “This study adds to the literature supporting triplet regimens — the combination is one of the most highly active and well-tolerated in MCL.”
MAVO: Acalabrutinib + Venetoclax + Obinutuzumab
Phase I/II (NCT04855695)
Populations
- R/R MCL (n=18)
- Treatment-naïve (n=21)
Results - R/R MCL
- ORR: 83%, CR: 78%
- 18-month PFS: 77%
Results - Treatment-Naïve
- ORR: 86%, CR: 81%
- TP53 aberrant (n=8): 88% ORR
- MRD-negative CR: 86%
- TP53 MRD-neg CR: 91%
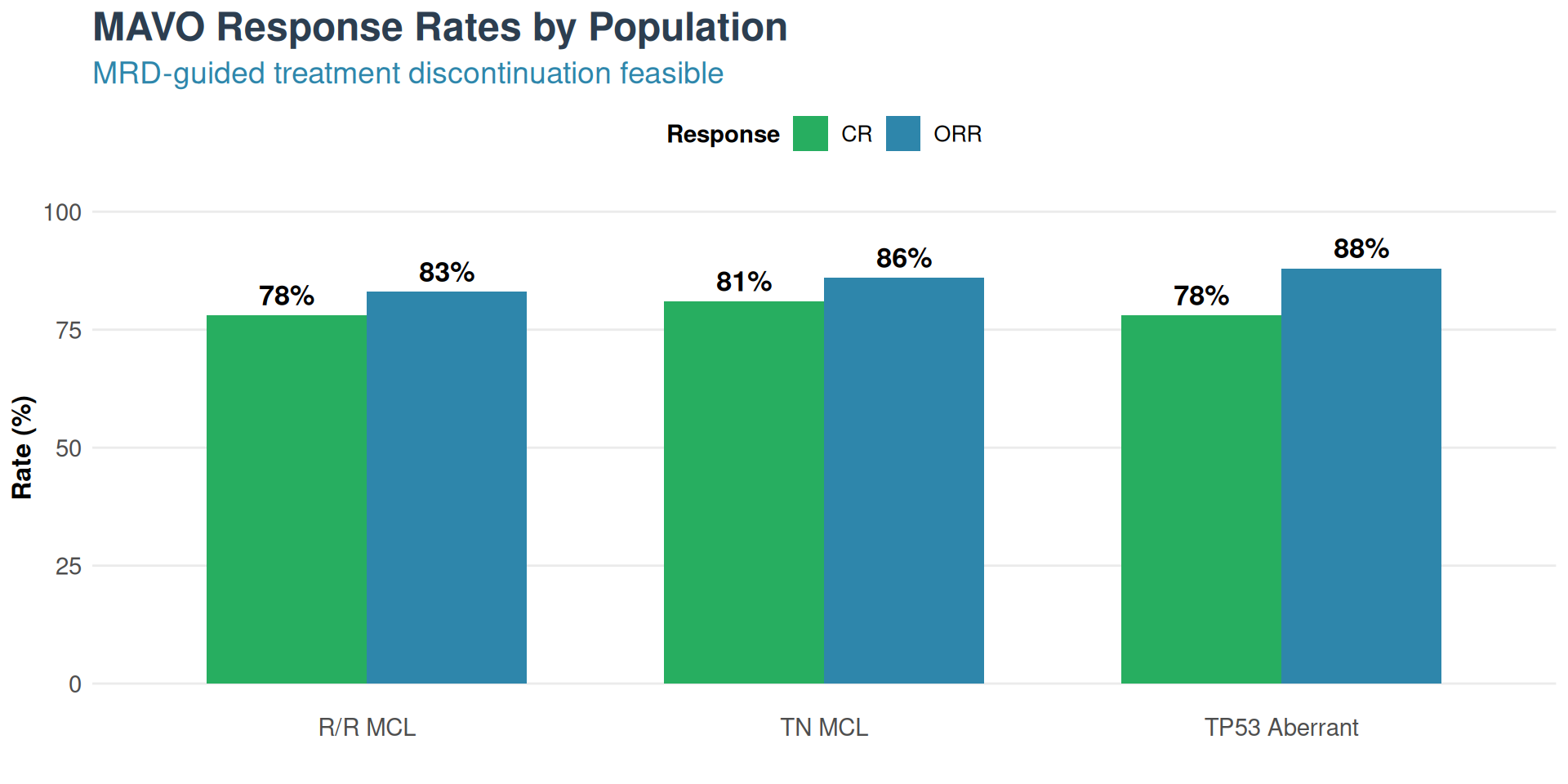
Pirtobrutinib: Non-covalent BTKi for cBTKi-refractory MCL
BRUIN study (NCT03740529)
Background
- C481S mutation in 50-75% of cBTKi progressors 1
- N=90 prior cBTKi patients
Mechanism
- Reversible binding away from C481
- Active against WT and C481S-mutant BTK
300-fold selectivity versus 98% of kinome
Results
- ORR: 57.8% (95% CI 46.9-68.1%)
- Median DOR: 21.6 months
- Historical post-BTKi survival: 2.9-8.4 months
Efficacy Results
| Metric | Value |
|---|---|
| ORR | 57.8% |
| CR | 20% |
| Median DOR | 21.6 months |
Pirtobrutinib: Safety & Regulatory
Safety Profile
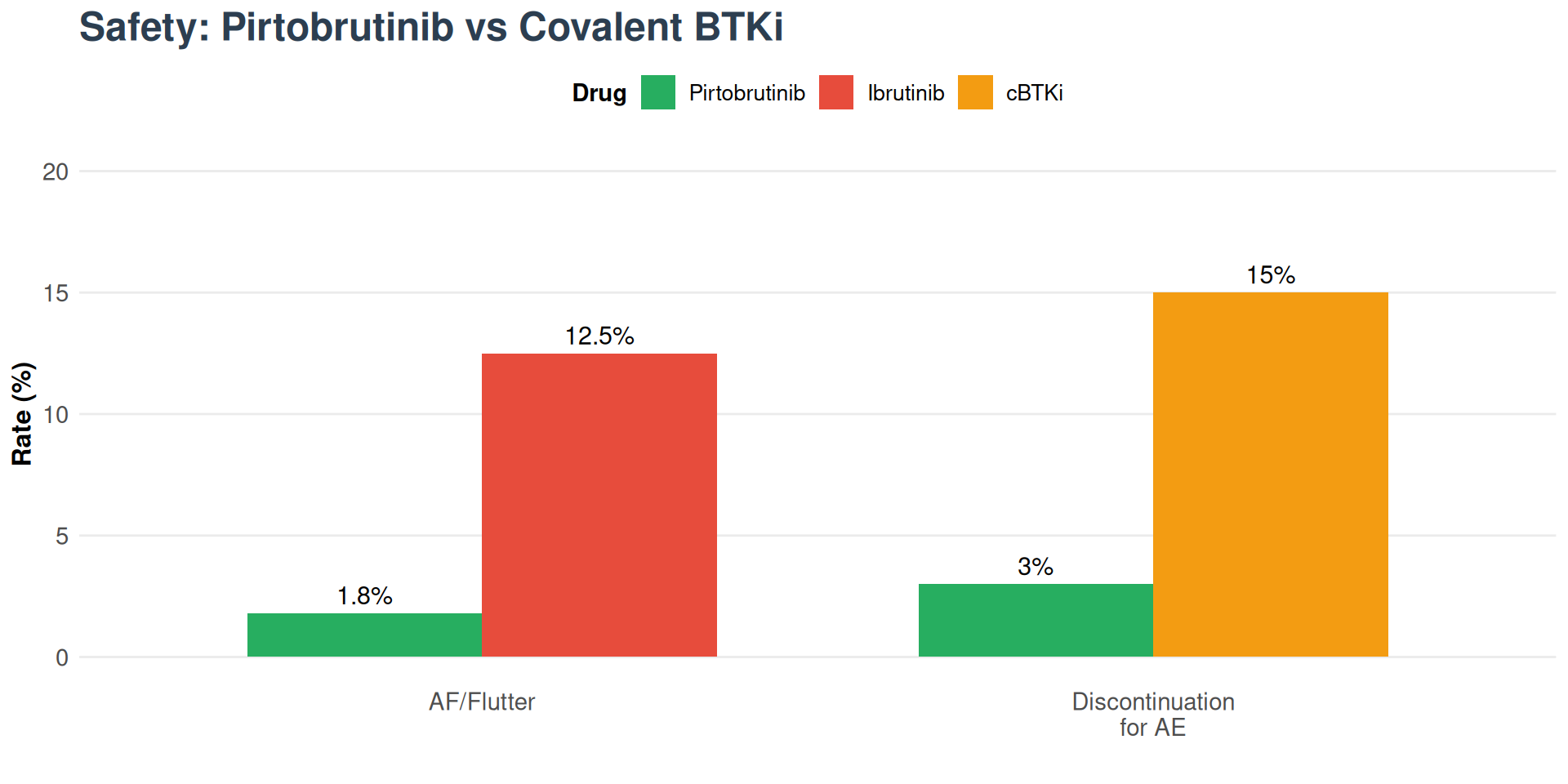
Regulatory Status
FDA Full Approval
December 3, 2025 - Converted from accelerated approval (Dec 2023)
- First-in-class non-covalent BTKi
- Phase III BRUIN MCL-321 ongoing (vs cBTKi in BTKi-naïve R/R MCL)
Waldenström Activity
- MRR: 71.3% in prior cBTKi patients
- MRR: 66.7% in cBTKi-refractory
- Off-label NCCN support for prior-treated WM
Expert Opinions on Pirtobrutinib
Clinical Investigators
- Michael Wang, MD from MD Anderson Cancer Center said:
- “Pirtobrutinib is really active in MCL — it is even effective after covalent BTKi resistance.”
- On safety: “Some patients asked whether they were in the placebo group due to minimal side effects.”
- Jennifer A. Woyach, MD from Ohio State University said:
- “The adverse effect profile is better with pirtobrutinib because it is such a selective molecule.”
Mechanism & Resistance
- Shuo Ma, MD, PhD from Northwestern University said:
- “Patients with C481S mutation can still respond to pirtobrutinib — it binds away from that site.”
- “The battle continues. We have to find new ways to fight resistance mechanisms.”
- Jeff Sharman, MD from Willamette Valley Cancer Institute said:
- “When covalent BTKi is no longer an option, pirtobrutinib extends the benefits of targeting the BTK pathway.”
- “It delayed subsequent treatment or death for 2.5 years in patients naive to venetoclax.”
Marginal Zone Lymphoma (MZL)
Agenda
Mantle Cell Lymphoma (MCL)
Marginal Zone Lymphoma (MZL)
Waldenström Macroglobulinemia (WM)
Clinical Implications
First CAR-T Approval and Emerging Therapies
Liso-cel: First CAR-T approved for MZL
TRANSCEND FL (NCT04245839)
Background
- MZL ~10% of NHL, 3 subtypes 1
- No prior disease-specific approved therapy
Method
- CD19 CAR-T, 1:1 CD4:CD8 composition
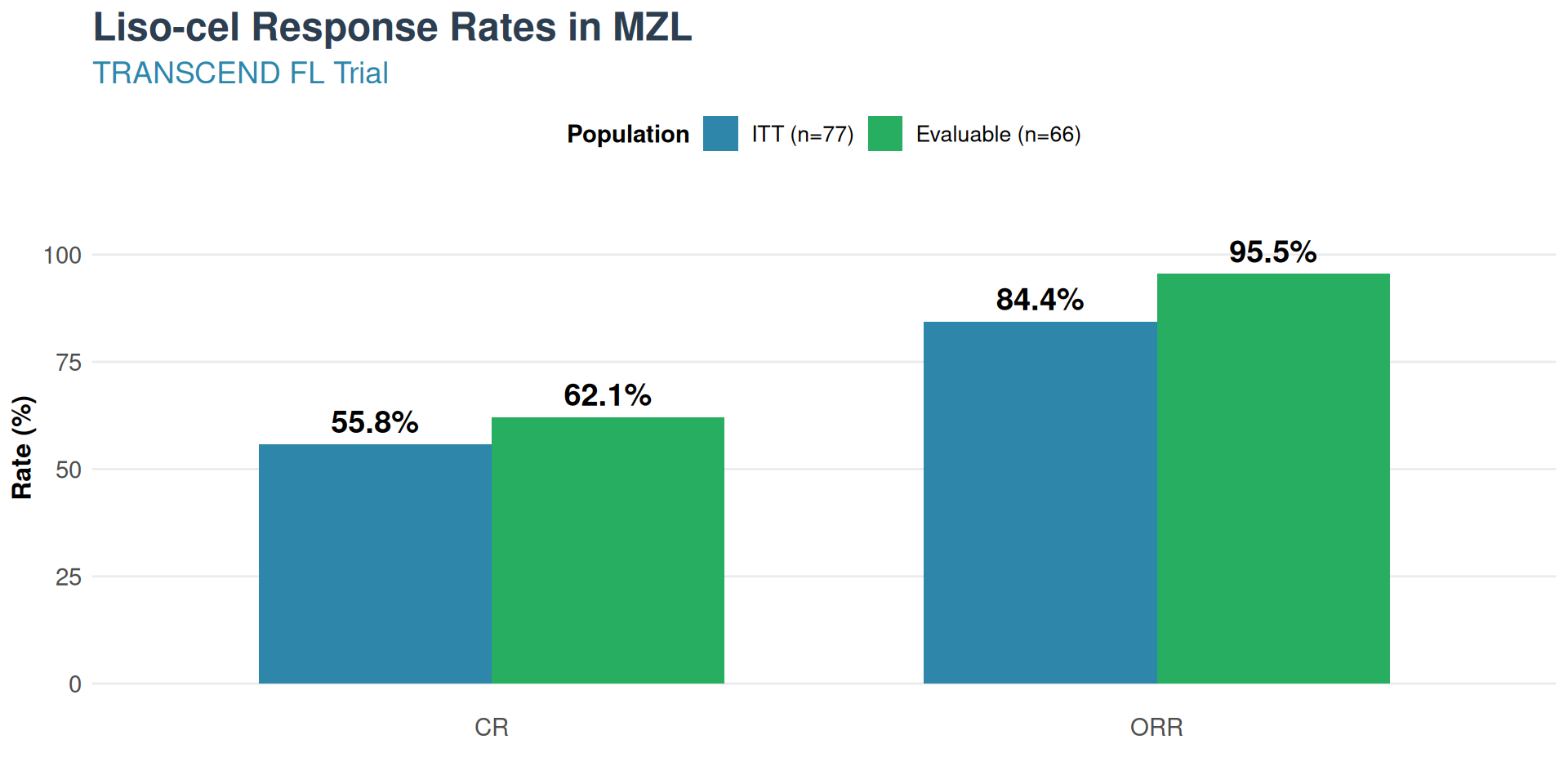
- N=77 (ITT) / 66 (evaluable)
Results
- ORR: 84.4% (ITT) / 95.5% (evaluable)
- CR: 55.8% (ITT) / 62.1% (evaluable)
- 24-month DOR: 88.6% maintained
- 24-month PFS: 85.7%
- 24-month OS: 90.4%
Liso-cel: Safety & FDA Approval
Safety Profile
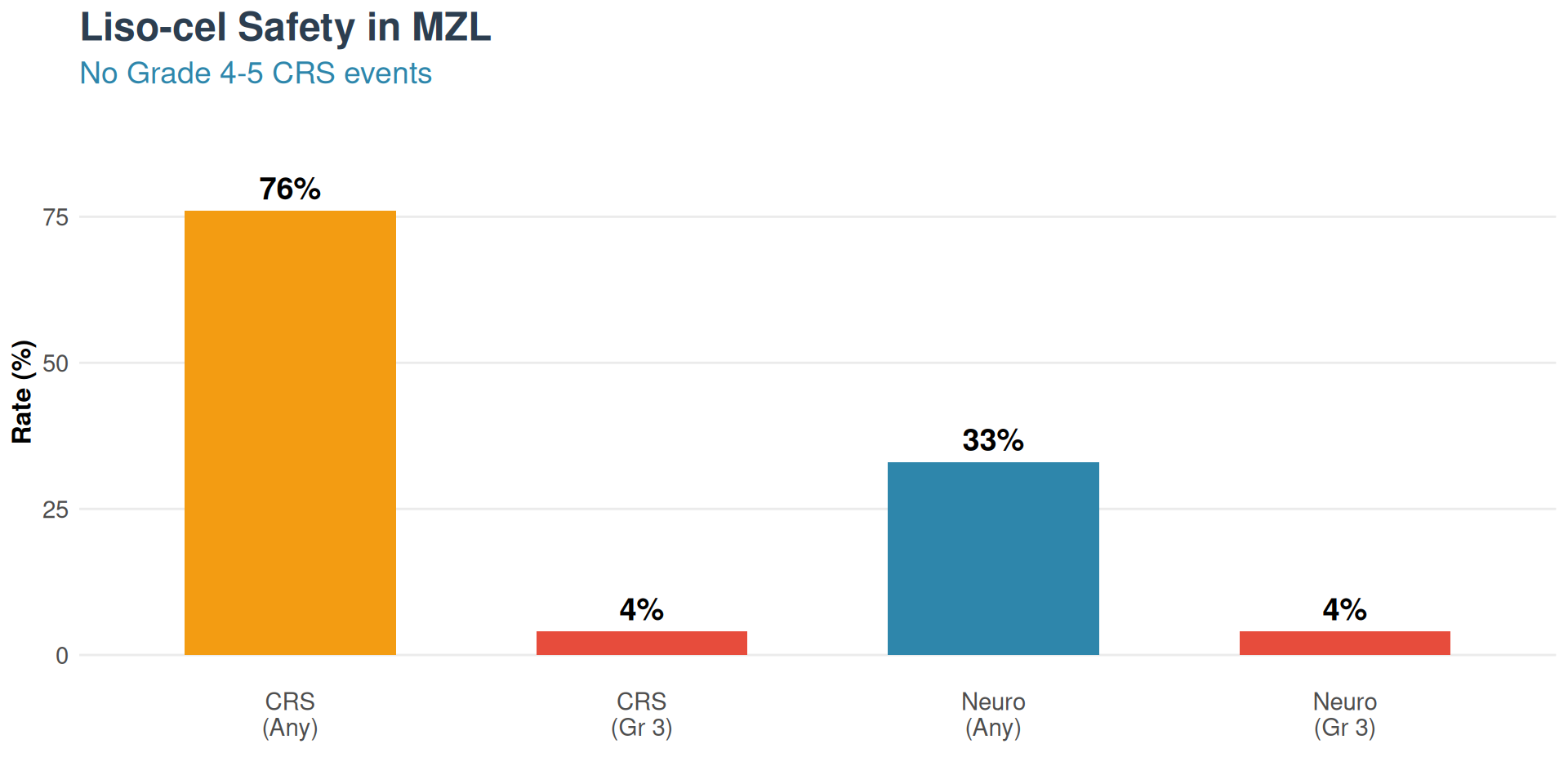
Regulatory Status
FDA Approval: December 4, 2025
First CAR-T therapy approved for MZL
- Fifth cancer indication for Breyanzi
- Most approved indications of any CD19 CAR-T
- Requirement: ≥2 prior lines of systemic therapy
- Orphan drug designation granted
Predictable Pharmacokinetics
- Unique 1:1 CD4:CD8 defined composition
- Consistent safety across all liso-cel indications
- Lower severe neurotoxicity than other CAR-T products
Expert Opinions on Liso-cel for MZL
Clinical Experts
- M. Lia Palomba, MD from Memorial Sloan Kettering Cancer Center said:
- “Median survival for MZL patients with multiple relapses is only 3 to 5 years — there was an urgent need for transformative therapies.”
- “Liso-cel is a critical advance and the first CAR T approved to treat MZL.”
- Manali Kamdar, MD from University of Colorado said:
- “The benefit in patients with high tumor burden and early progressing disease is just spectacular.”
Regulatory & Industry
- Vinay Prasad, MD from FDA said:
- “This represents a major advancement in precision medicine, turning the patient’s immune system into a more effective tool.”
- Lynelle B. Hoch from Bristol Myers Squibb said:
- “Breyanzi is now the leading CD19-directed CAR T therapy covering the broadest range of B-cell malignancies — five cancer types.”
- Rosanna Ricafort, MD from Bristol Myers Squibb said:
- “For patients who relapse, the disease can be quite aggressive. The 1:1 CD4:CD8 composition provides predictable pharmacokinetics.”
BGB-16673: BTK Degrader for BTKi-Resistant Disease
CaDAnCe-101 Trial (NCT05006716)
Mechanism
- CDAC recruits cereblon E3 ligase
- Induces BTK ubiquitination and proteasomal degradation
- Eliminates entire BTK protein (not just inhibition)
Targets
- C481S mutations
- “Kinase-dead” scaffold mutations (L528W)
- Active regardless of prior BTKi exposure
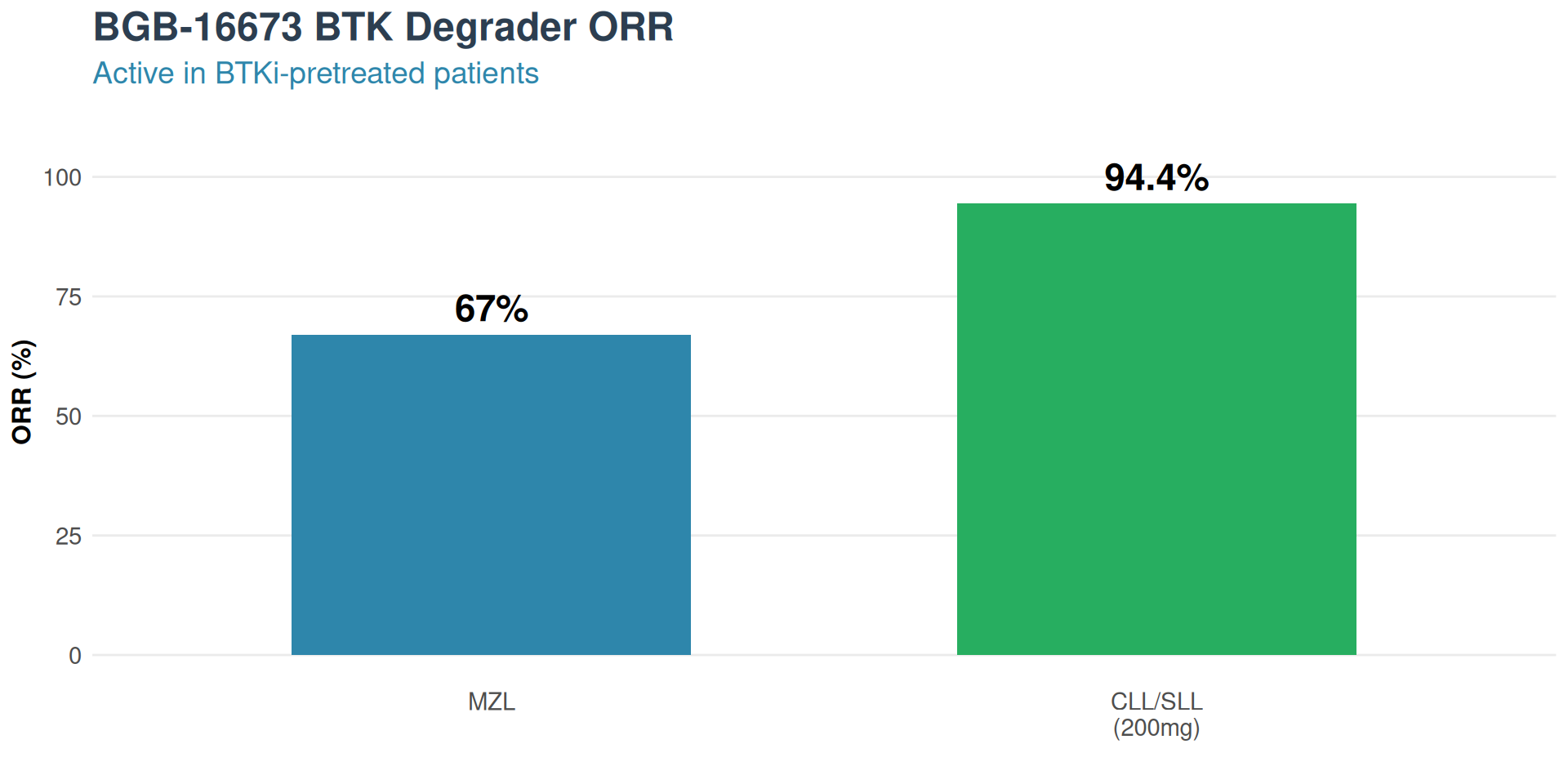
Results
- MZL ORR: 67% in response-evaluable
- CLL/SLL ORR: 94.4% at 200mg dose (RDFE)
Phase 3 trial activated: BGB-16673 vs Pirtobrutinib head-to-head
Expert Opinions on BGB-16673 BTK Degrader
Mechanism Experts
- Lydia Scarfo, MD from Università Vita-Salute San Raffaele said:
- “The greatest advantage is targeting BTK for degradation regardless of mutation status — it works on both wild-type and C481S-mutated protein.”
- Chan Cheah, MBBS from Linear Clinical Research (Perth) said:
- “Rather than being a small molecule inhibitor, it’s got a bifunctional domain which causes degradation via polyubiquitination.”
- “They retain efficacy in both covalent and non-covalent BTKi-exposed patients.”
Clinical Investigators
- Meghan Thompson, MD from Memorial Sloan Kettering said:
- “CaDAnCe-101 data shows a tolerable safety profile with responses in heavily pretreated patients.”
- In-hye Ahn, MD from Dana-Farber Cancer Institute said:
- “The drug tags BTK for ubiquitination, leading to complete destruction by the cell’s own proteasome machinery.”
- “A phase 3 head-to-head trial vs pirtobrutinib has already been activated.”
Evorpacept: CD47 Blockade + R² for Frontline iNHL
Phase II (ASH 2025)
Mechanism
- Blocks CD47-SIRPα “don’t eat me” signal
- Enables antibody-dependent cellular phagocytosis
Population
- N=24 (14 FL, 10 MZL)
- Previously untreated indolent NHL
Method
- Evorpacept + Rituximab + Lenalidomide (R²)
Results
- CR rate: 92% (vs ~50% historical R²)
- ORR: 100%
- 1-year PFS: 91%
- 1-year OS: 100%
Evorpacept + R² vs Historical R²
| Metric | Evorpacept + R² | R² Historical |
|---|---|---|
| CR | 92% | 50% |
| ORR | 100% | 80% |
| 1-year PFS | 91% | 75% |
| 1-year OS | 100% | 95% |
Waldenström Macroglobulinemia (WM)
Agenda
Mantle Cell Lymphoma (MCL)
Marginal Zone Lymphoma (MZL)
Waldenström Macroglobulinemia (WM)
Clinical Implications
Fixed-Duration Therapy and High-Risk Strategies
Pirtobrutinib + Venetoclax: Fixed-Duration WM Therapy
Dana-Farber Phase II
Background
- WM typically requires indefinite BTKi therapy
- Cumulative toxicities and resistance concerns
Population
- N=27 evaluable (MYD88-mutant)
- Previously treated, symptomatic
Method
- Fixed 24-cycle oral combination
Results
- ORR: 100%
- CR/VGPR: 56% (exceeds futility threshold)
- MRR: 87%
- Median time to VGPR: 1.9 months
- No arrhythmia events to date
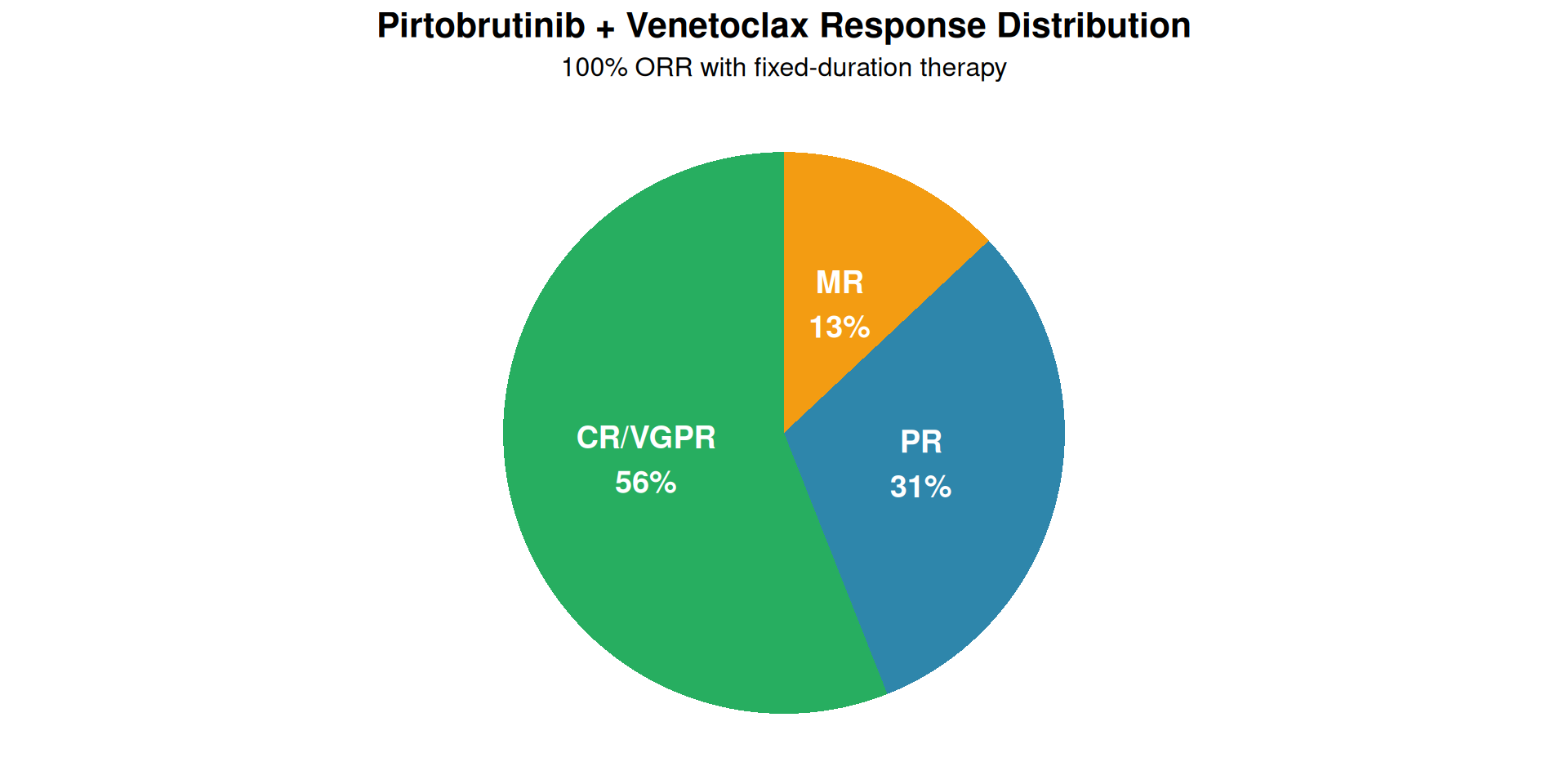
“Depth of response beyond what was expected for each medication separately” - Dr. Jorge Castillo
Loncastuximab Tesirine: ADC for High-Risk WM
Phase II Trial
Background
- CD19-targeting antibody-drug conjugate
- N=14, median 4 prior therapies
- High-risk: 8 CXCR4+, 8 TP53-altered
Method
- Fixed 6-dose regimen
- 150 µg/kg cycles 1-2, 75 µg/kg cycles 3-6
Results
- Overall ORR: 86%
- CR/VGPR: 71%
- TP53-altered: 100% response, 89% CR/VGPR
- Active in CXCR4-mutant (BTKi-resistant)
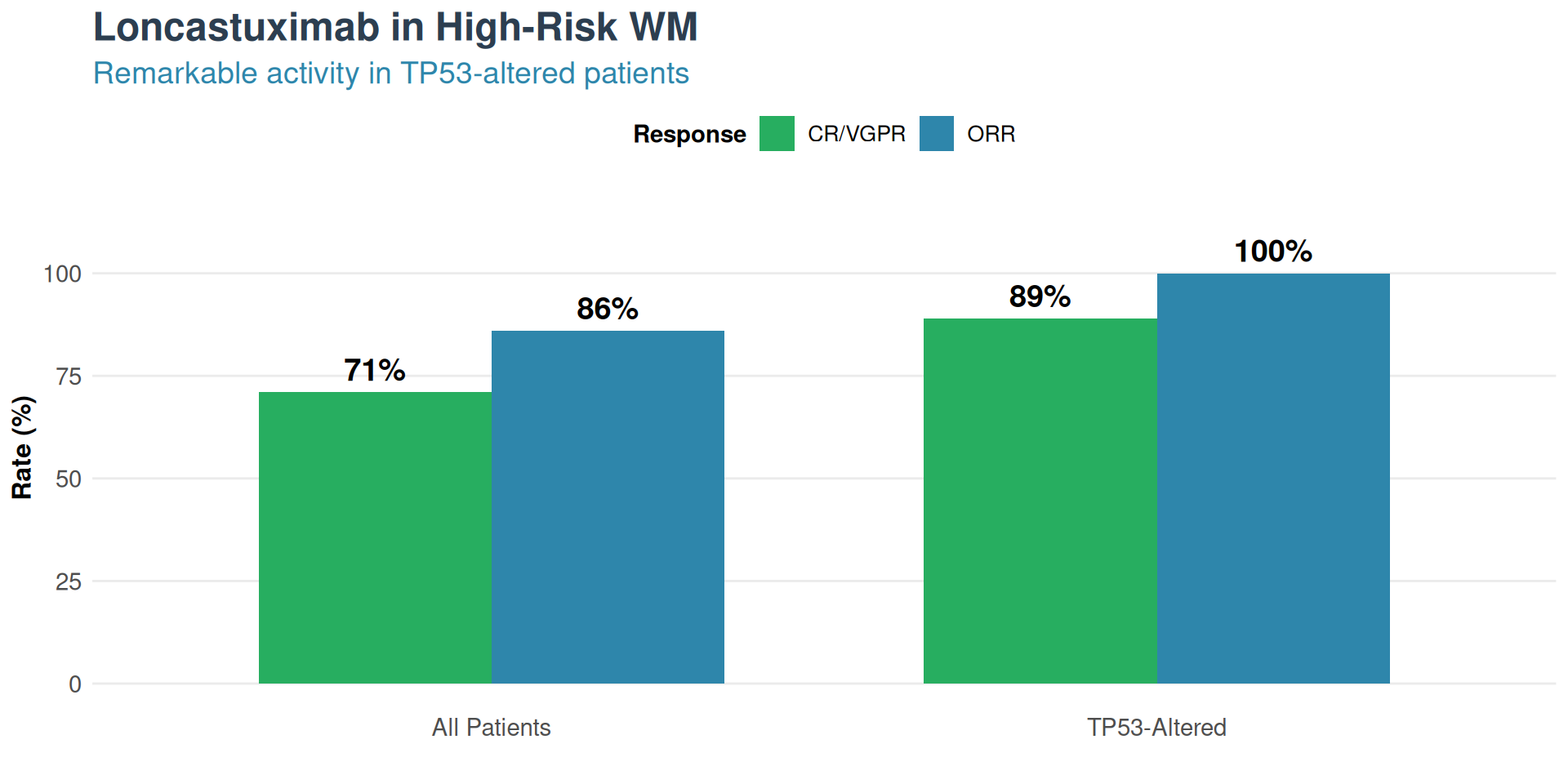
Note: Loncastuximab FDA-approved for DLBCL, not yet for WM
Expert Opinions on WM Fixed-Duration Therapy
Lead Investigators
- Jorge Castillo, MD from Dana-Farber Cancer Institute said:
- “The depth of response is beyond what was expected for each medication separately — three of 27 patients attained CR in the first six months.”
- “Our goal is to provide patients with a treatment that is non-chemotherapy, fixed duration, and all oral.”
- Steven Treon, MD from Dana-Farber said:
- “For patients with acquired resistance to covalent BTK inhibitors, newer options include pirtobrutinib or venetoclax.”
Treatment Perspectives
- Christian Buske, MD from University Hospital Ulm said:
- “The trend goes towards chemotherapy-free approaches as fixed-duration treatment for one or two years.”
- Shayna Sarosiek, MD from Dana-Farber said:
- “Emerging treatments such as BTKi combinations, BCL2 antagonists, BTK degraders, and immunotherapy are under investigation.”
- “No arrhythmia events reported to date — addressing a key concern with long-term BTKi use.”
Clinical Implications
Agenda
Mantle Cell Lymphoma (MCL)
Marginal Zone Lymphoma (MZL)
Waldenström Macroglobulinemia (WM)
Clinical Implications
Key Takeaways from ASH 2025
1. End of One-Size-Fits-All
- Molecular testing at diagnosis now determines optimal therapy
- TP53-mutant MCL → BOVen (not chemoimmunotherapy)
- MYD88 wild-type WM → Different BTKi approach
- CXCR4 mutations → Consider resistance patterns
2. Resistance Mechanisms Guide Sequencing
- C481S mutation → Pirtobrutinib effective
- BTK degraders → Target scaffold mutations
- Rational therapy sequencing now possible
3. CAR-T Manufacturing Innovation
- GLPG5101: 7-day vein-to-vein time
- May eliminate 15-20% dropout rates
- Technology validated despite commercial uncertainty
4. MZL Milestone
- Liso-cel: First disease-specific approved therapy
- 95.5% ORR in evaluable patients
- Durable responses (88.6% at 24 months)
5. Fixed-Duration Therapies
- Pirtobrutinib + Venetoclax in WM: 100% ORR
- May replace indefinite BTKi therapy
- MRD-guided discontinuation feasible
6. Novel Mechanisms
- BTK degraders (BGB-16673): Beyond inhibition
- CD47 blockade (Evorpacept): 92% CR with R²
- New targets for resistant disease
References
Key Trials
- ATALANTA-1: ASH 2025 Abstract #662, Galapagos NV
- ECHO Trial: NCT02972840, FDA approval January 16, 2025
- BOVen: Blood 2025;145(5):497, NCT03824483
- MAVO: Blood 2024;144(Suppl 1):4408, NCT04855695
- BRUIN: JCO 2023, NCT03740529, FDA full approval December 2025
- TRANSCEND FL MZL: NCT04245839, FDA approval December 4, 2025
Additional Sources
- FDA.gov Drug Approvals
- ClinicalTrials.gov
- ASH Abstract Database
- NCCN Guidelines (accessed December 2025)
- Dana-Farber Cancer Institute Press Releases
- Galapagos NV Press Releases
Data Validation Notes
- TRANSCEND FL MZL ORR: 84.4% (ITT) vs 95.5% (evaluable) - both correct for different analysis sets
- Pirtobrutinib MCL ORR varies 50-57.8% depending on data cutoff and cohort definition
- BGB-16673 is a BTK degrader/CDAC, not technically a classic PROTAC
- SWOG S2005 (WM) has no efficacy results - trial ongoing
ASH 2025 | B-Cell Lymphoma Updates